Abstract
The aqueous and chloroform fractions of the methanolic extract obtained from the aerial parts of Saponaria officinalis L. were evaluated for their antibacterial and antifungal activities. The aqueous fraction exhibited the highest antibacterial activity, with inhibition zones measuring 12 mm against Bacillus subtilis, 13 mm against Staphylococcus aureus, 18 mm against Escherichia coli, and 14 mm against Pseudomonas aeruginosa. In silico PASS predictions indicated that sapolide, a major constituent, possesses high probabilities of biological activity, including 92.4% for general antineoplastic effects, 88.7% for pro-apoptotic activity, and 85.4% for antineoplastic activity against lung cancer. Pharmacokinetic analysis revealed that sapolide complies with Lipinski’s “rule of five,” shows good oral bioavailability, and meets the criteria for drug-likeness. Molecular docking analysis revealed that sapolide binds to Aurora B kinase with binding energies ranging from –10.7 to –7.4 kcal/mol, and to the Pregnane X Receptor with energies from –8.8 to –7.5 kcal/mol. These interactions suggest stable and specific binding, with the sapolide–Aurora B kinase complex, in particular, supporting its potential as a novel inhibitor of this enzyme with antitumor activity. In summary, these findings highlight sapolide as a promising bioactive natural compound with both antimicrobial effects and significant therapeutic potential in oncology.
Full article
INTRODUCTION
The genus Saponaria (Caryophyllaceae) comprises about 40 species distributed across the temperate regions of Eurasia, with a center of diversity in the Mediterranean area [1]. Six species are endemic to the territory of Uzbekistan. Among them, Saponaria officinalis L. is particularly notable for its exceptionally high saponin content, which constitutes 20–35% of the root dry mass. Phytochemical investigations of its roots have led to the isolation of various metabolites, including carbohydrates and triterpenoid glycosides such as saponazides [2] and saponariosides A–M [3-5] together with four aglycones: hederagenin, hydroxyhederagenin, gypsogenin, and quillaic acid [6]. In addition, the leaves of S. officinalis contain alkaloids, ascorbic acid, and flavonoids such as vitexin, saponarin, and saponaretin [7,8]. In traditional medicine, decoctions and infusions prepared from the roots and leaves of S. officinalis have long been employed for the treatment of respiratory diseases, gastrointestinal disorders, rheumatism, polyarthritis, psoriasis, lichen planus, eczema, and prostate adenoma [9,10]. Recent phytochemical studies of the aerial parts of S. officinalis growing in Uzbekistan resulted in the isolation of a new natural saponin, sapolide [11]. This compound had previously only been obtained synthetically through acid hydrolysis of albigenic acid [12]. In modern pharmaceutical research, computer-aided drug design (CADD) has become an essential tool for the discovery of new therapeutic agents with improved efficacy and safety profiles [14]. Among the most widely applied strategies are structure-based drug design (SBDD) and ligand-based drug design (LBDD), which allow the evaluation of biological activity, toxicity, physicochemical parameters, and pharmacokinetic properties of candidate molecules [15].
The present study we evaluate the antibacterial and antifungal activities of extracts and fractions from S. officinalis and perform an in silico investigation of sapolide, focusing on its predicted biological activity, safety, solubility, bioavailability, pharmacokinetics, and drug-likeness.
MATERIALS AND METHODS
Plant material. The aerial parts of Saponaria officinalis were used in this study. The plant material was collected in July 2020 during the full flowering stage in the Yunusabad district of Tashkent, Uzbekistan. The samples were air-dried, packed in paper bags, and stored in a cool, dark place until further use. Extraction and isolation. The powdered air-dried aerial parts of the plant (1 kg) were extracted five times with methanol at room temperature. After vacuum evaporation, a crude extract (445 g) was obtained, which was suspended in water (5 L) and successively fractionated with chloroform, ethyl acetate, and n-butanol.
The n-butanol extract (120 g) was chromatographed on a silica gel column (0.03–0.200 mm) using a gradient solvent system of CHCl₃–CH₃OH (1:0 → 0:1), yielding four fractions (A–D). Fraction 1 (56 g) was rechromatographed on silica gel (0.040–0.063 mm) with a gradient system of CHCl₃–CH₃OH (50:1 → 0:1), resulting in ten subfractions (A1.1–A1.10). Subfraction A1.1–5–6 (48 mg) was further purified by preparative TLC (CHCl₃–CH₃OH, 40:1), affording pure sapolide (20 mg). The yield was 0.002% relative to the air-dried plant material. Determination of antimicrobial activity. The fractions of the methanolic extract of Saponaria officinalis were evaluated for antibacterial and antifungal activities using a modified agar diffusion method [13].
In silico analysis of sapolide. The biological activity of sapolide was evaluated using the PASS Online, SwissADME, and SwissTarget programs. PASS Online (https://www.way2drug.com) was used to predict the biological activity of sapolide based on structure–activity relationship analysis [16]. SwissADME (SIB, www.expasy.org/resources/swissadme) was used to evaluate the physicochemical properties, solubility, bioavailability, and pharmacokinetic parameters. SwissTarget (SIB, www.expasy.org/resources/swisstargetprediction) was used to predict potential protein targets. Molecular docking was performed using AutoDock Vina 4.3, which was downloaded from the official website (www.scripps.edu). The AutoDock tools were used to generate the PDBQT file. All docking calculations were carried out with the AutoDock Vina 4.3 software package in combination with auxiliary tools AutoDock Tools and PyMOL. The crystalline structures of macromolecules were obtained from the RCSB Protein Data Bank (www.rcsb.org/pdb/). All calculations were performed in AutoDock Vina 4.3 using AutoDock Tools and PyMOL.
RESULTS AND DISCUSSION
Determination of antimicrobial and antifungal activity. The results of the antimicrobial activity of Saponaria officinalis extracts are presented in Table 1.
The results (Table 1) showed that the tested extracts inhibited the growth of both Gram-positive and Gram-negative bacterial test strains. The aqueous fraction exhibited the highest antibacterial activity, with inhibition zones measuring 12 mm for Bacillus subtilis, 13 mm for Staphylococcus aureus, 18 mm for Escherichia coli, and 14 mm for Pseudomonas aeruginosa.
In silico study of sapolide. The in silico predictions of the biological activity of sapolide were obtained using the PASS Online program by inputting the chemical structure of the compound. The results were considered within. According to the predictions of the PASS Online service (Table 1), the compound sapolide demonstrated the highest probabilities of activity: 0.924 (92.4%) for antineoplastic activity, 0.887 (88.7%) for apoptosis agonist properties, and 0.854 (85.4%) for antineoplastic (lung cancer) activity.
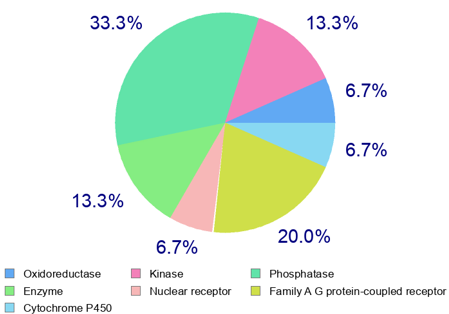
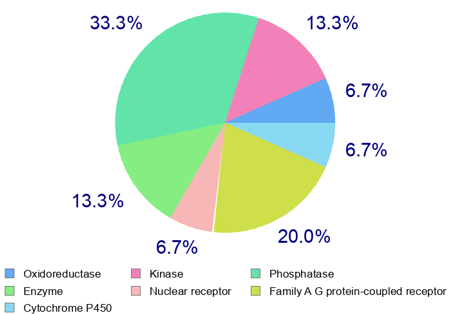
The prediction of physicochemical properties, pharmacokinetic parameters, and drug-likeness indicators of sapolide was carried out using the SwissADME program (Table3). Based on the results obtained using the SwissADME program (Table 2), it can be predicted that sapolide meets the pharmacokinetic criteria (absorption, distribution, metabolism, and excretion) as well as the physicochemical requirements (no more than five hydrogen bond donors, ten hydrogen bond acceptors, and a molecular weight not exceeding 500 Da, among others) defined by Lipinski’s “Rule of Five.” The compound demonstrates good bioavailability and satisfies the criteria for drug-likeness. The SwissTarget program compares the chemical structure of the studied compound with a database of known molecules and predicts similar interactions based on this comparison. Using the SwissTarget program, various macromolecules that may serve as potential biological targets for sapolide were predicted (Figure 1). As shown in Figure 1, the results obtained from the SwissTarget program predict that sapolide interacts with macromolecules in the following order: oxidoreductases (6.7%), kinases (13.3%), phosphatases (33.3%), and so on. Molecular docking. Using AutoDock Vina 4.3, molecular docking was performed to predict the binding mode and affinity of the sapolide ligand to the biological targets—macromolecules identified with the SwissTarget program. AutoDock Vina 4.3 predicts the optimal spatial orientation of the ligand and calculates the interaction energy between the ligand and the macromolecule. As shown in Figure 2, two oxygen-containing fragments at positions 16 and 28 are involved in interactions with TYR A:141 (interaction between the carbonyl group of sapolide and the hydroxyl group of tyrosine) and with GLN D:858 (interaction between the hydroxyl group of sapolide and the glutamine residue of the protein). The blue areas indicate potential regions of electron density, representing electrostatic interactions that correspond to hydrogen bonds. Green dashed lines depict hydrogen bonds between the sapolide ligand and the amino acid residues of Aurora B kinase. The residues shown in green circles are amino acids located approximately 4–5 Å from the ligand, indicating possible hydrophobic, π–π, van der Waals, and other interactions.
Molecular docking determines the binding affinity and potential biological activity of a low-molecular-weight compound. In docking analysis, it is generally accepted that a ligand with a lower (more negative) binding energy to a macromolecule indicates stronger potential binding [17]. The active binding sites of the Aurora B kinase protein are as follows:
• Hydrophobic: LEU D:865, PRO D:862, ILE D:855, PHE A:101, PRO A:336/337/158, TYR A:156
• Polar/charged: HIS A:133/134, ARG A:139, LYS A:215, GLU A:155, SER A:338
1. Hydrogen bonds (green dashed lines): Classical hydrogen bonds are formed between the ligand molecule and the following amino acid residues of the protein: GLN (D:858), TYR (A:141).
2. Van der Waals interactions (light green lines): The ligand molecule establishes weak Van der Waals interactions with multiple amino acid residues of Aurora B kinase. These interactions are observed with the following amino acids: ILE (D:855), TYR (D:859), PHE (A:101), GLU (A:155), TYR (A:156), PRO (A:158), LEU (A:210), GLY (A:209), LYS (A:215), PRO (A:135, A:336, A:337, D:862, D:863), LEU (D:865), HIS (A:133, A:134), SER (A:338). These weak hydrophobic interactions contribute to the proper positioning of the ligand within the active site and enhance the overall stability of the binding.
1. From Figure 3, the following types of interactions can be identified:
2. Hydrogen bonds (green dashed lines): Classical hydrogen bonds are formed between the hydrophilic groups of the ligand and the residues ARG (A:173), LYS (A:170), and HIS (A:168) of the PXR protein. These bonds enhance the binding selectivity and stability of the complex, stabilizing the ligand within the active site of the macromolecule.
3. π-Alkyl interactions (purple dashed lines): π-Alkyl interactions occur between the aromatic rings of the ligand and the residues LYS (A:252) and TYR (A:249) of the protein. The interaction of aromatic residues with the saturated regions of the ligand further enhances the stability of the complex.
4. C–H or Van der Waals interactions (light green lines):
The ligand molecule forms weak Van der Waals interactions with several amino acid residues of the Pregnane X Receptor (PXR, PDB: 8SVN). These interactions are observed with the residues ASN (A:171), PHE (A:169), PHE (A:172), THR (A:248), THR (A:422), and PRO (A:423). These weak interactions contribute to the proper positioning of the ligand within the active site and help maintain its stability. A carbon–hydrogen bond is observed between the ligand and the residue ASP (A:245). This weak interaction further stabilizes the position of the ligand.
CONCLUSION
The aqueous and chloroform fractions of the methanolic extract of the aerial parts of Saponaria officinalis L. exhibited antimicrobial activity, with the aqueous fraction showing the strongest inhibition against both Gram-positive and Gram-negative bacterial strains. This effect is most likely associated with the high content of saponins. The triterpenoid glycoside sapolide, isolated from this plant, demonstrated promising pharmacological potential in silico. PASS predictions indicated high probabilities for antineoplastic activity, apoptosis agonist properties, and activity against lung cancer. Pharmacokinetic and physicochemical analyses confirmed its compliance with Lipinski’s “Rule of Five,” good oral bioavailability, and overall drug-likeness. Molecular docking revealed strong and stable binding of sapolide to Aurora B kinase and the Pregnane X receptor, with binding energies in the range of –10.7 to –7.4 kcal/mol and –8.8 to –7.5 kcal/mol, respectively. These interactions suggest that sapolide may serve as a promising ligand and potential inhibitor of Aurora B kinase, supporting its further development as a candidate for antitumor therapy. Overall, these findings highlight sapolide as a bioactive natural compound with dual antimicrobial and anticancer potential, warranting further in vitro and in vivo investigations.
Figures
Keywords
References
1.Dashti, A., (2018). Role of seed micro-morphology in the taxonomy of Saponaria (Caryophyllaceae). Iranian Journal of Botany, 24(2), 130–137. 10.22092/ijb.2018.123401.1213.
2.Satish, Ch., Dharmendra, S Rawat., Arun, Bhatt., Phytochemistry and pharmacological activities of Saponaria officinalis L.: A review. (2021). Notulae Scientia Biologicae 13(1), 10809. https://doi.org/10.15835/nsb13110809.
3.Koike, K., Jia, Z., & Nikaido, T. (1999). New triterpenoid saponins and sapogenins from Saponaria officinalis. Journal of Natural Products, 62(12), 1655–1659. https://doi.org/10.1021/np990311r.
4.Jia, Z., Koike, K., & Nikaido, T. (1998). Major triterpenoid saponins from Saponaria officinalis. Journal of Natural Products, 61(11), 1368-1373. https://doi.org/10.1021/np980167u.
5.Jia, Z., Koike, K., & Nikaido, T. (1999). Saponarioside C, the first α-D-galactose containing triterpenoid saponin, and five related compounds from Saponaria officinalis. Journal of Natural Products, 62(4), 449–453. https://doi.org/10.1021/np980434w.
6.Smułek, W., Zdarta, A., Pacholak, A., Zgoła-Grześkowiak, A., Marczak, Ł., Jarzębski, M., & Kaczorek, E. (2017). Saponaria officinalis L. extract: Surface active properties and impact on environmental bacterial strains. Colloids and Surfaces B: Biointerfaces, 150, 209-215. https://doi.org/10.1016/j.colsurfb.2016.11.035.
7.Endonova, G. B., Antsupova, T. P., & Zhamsaranova, S. D. (2015). Study of Flavonoid and Antioxidant Activity of Saponaria officinalis. Biosciences Biotechnology Research Asia. 12(3), 2017-2021. http://dx.doi.org/10.13005/bbra/1869.
8.Lu, Y., Van, D., Deibert, L., Bishop, G., Balsevich, J. (2015). Antiproliferative quillaic acid and gypsogenin saponins from Saponaria officinalis L. roots. Phytochemistry 113, 108–120. https://doi.org/10.1016/j.phytochem.2014.11.021
9.Charalambous, D., Christoforou, M., Christou, K., Christou, M., Ververis, A., Andreou, M., & Pantelidou, M. (2024). Saponin and phenolic composition and assessment of biological activities of Saponaria officinalis L. root extracts. Plants, 13(14). https://www.mdpi.com/2223-7747/13/14/1982.
10.Ashurova, L. N., Bobakulov, Kh. M., Ramazonov, N. Sh., Sasmakov, S. A., Ashirov, O. N., Azimova, Sh. S., & Abdullaev, N. D. (2021). Essential oil from the aerial part of Saponaria griffithiana and Saponaria officinalis. Chemistry of Natural Compounds, 57(5), 970–972. https://doi.org/10.1007/s10600-021-03527-3.
11.Kubota, T., Kitatani, H., & Hinoh, H. (1969). Isomerisation of quillaic acid and echinocystic acid with hydrochloric acid. Tetrahedron Letters, 10, 771-774. https://doi.org/10.1016/S0040-4039(01)87805-9.
12.Wayne, P. A. (2009). Performance Standards for Antimicrobial Susceptibility Testing; Nineteenth Informational Supplement (CLSI document M100-S19). Clinical and Laboratory Standards Institute, Wayne, PA, USA.
13.Vasiliev, P. M., Golubeva, A. V., Koroleva, A. R., Perfiliev, M. A., & Kochetkov, A. N. (2023). Title of the article. Safety and Risk of Pharmacotherapy, 11(4), 390–408. https://doi.org/10.30895/2312-7821-2023-11-4-390-408.
14.Garaev, E. A., Huseynguliyeva, K. F. (2021). Modern approaches to assessing the toxicity of xenobiotics. Azerbaijan Medical Journal, 2, 95–100.
15.D.A. Filimonov, A.A. Lagunin, T.A. Gloriozova, A.V. Rudik, D.S. Druzhilovsky, P.V. Pogodin, V.V. Poroykov, Chemistry of Heterocyclic Compounds, 3, 483–499 (2014).
16.K.V. Goldaeva, Journal of Bioinformatics and Genomics, 4(26) (2024).
https://doi.org/10.60797/jbg.2024.26.6
Article Info:
Publication history
Published: 07.Apr.2026
Copyright
© 2022-2025. Azerbaijan Medical University. E-Journal is published by "Uptodate in Medicine" health sciences publishing. All rights reserved.Related Articles
IN SILICO STUDY TAURINE DEHYDROGENASE INHIBITOR ACTIVITY OF SOME ARYL-HYDRASONES OF α-KETOAETHERS
Viewed: 447



